CEL-SCI Corporation Reports Third Quarter Fiscal 2024 Financial Results
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20240815767286/en/
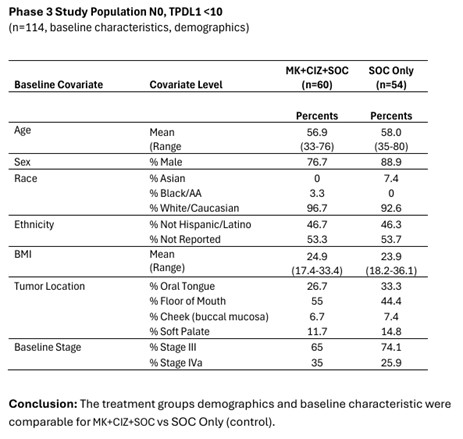
The table includes detailed results from the bias analysis. (Graphic: Business Wire)
Clinical and Corporate Developments include:
-
In
July 2024 , following the end of the third fiscal quarter,CEL-SCI reported the results of a bias analysis. The bias analysis, a standard process to ensure a trial’s findings are reliable, was conducted in preparation for CEL-SCI’s upcoming confirmatory Registration Study. This study will enroll 212 patients representing the target population for which Multikine had a 5-year survival of 73% vs. 45% in the control group. Detailed data on parameters including patient age, sex, race, tumor locations, and staging demonstrated balance between the treatment and control arms. No bias helping Multikine was found, supporting confidence that Multikine is the reason for the large increase in patient survival. -
CEL-SCI received the go-ahead from the FDA for its confirmatory Registration Study of Multikine in the treatment of head and neck cancer based on strong safety and efficacy data from the completed Phase 3 study which enrolled 928 patients. The FDA agreed with the pre-surgical selection of patients most likely to benefit from Multikine—those with newly diagnosed advanced primary head and neck cancer with no lymph node involvement (determined via PET scan) and with low PD-L1 tumor expression (determined via biopsy). In this target population in the Phase 3 study, Multikine demonstrated a 5-year survival of 73% vs a 45% survival in the control patients as well as a hazard ratio of 0.35. -
Dr.
Eyal Talor , CEL-SCI’s Chief Scientific Officer, delivered a presentation titled “Neoadjuvant Immunotherapy for Head and Neck Cancer: Low Tumor PD-L1 Expression - IT-MATTERS – RCT” at the International Drug Discovery Science & Technology (IDDST) 20th Annual Congress inBudapest, Hungary . Highlights of the presentation include:- Multikine significantly increases overall survival in patients with low levels of tumor cell PD-L1 expression in contrast to approved checkpoint inhibitors
- 73% survival for Multikine vs 45% in the control arm at 5 years
- Statistically significant log rank p = 0.0015
- 5-year risk of death cut in half from 55% to 27%
- Hazard ratio = 0.35 (95% CIs [0.19, 0.66])
- The presentation may be accessed on CEL-SCI’s website at the following LINK
-
CEL-SCI strengthened its Board of Directors with the addition ofMario Gobbo and the appointment ofRobert Watson as Chairperson of the Board.Mr. Gobbo has nearly 40 years of banking and corporate finance experience in healthcare and energy.Mr. Watson brings extensive expertise in capital formation strategies and partnerships, having negotiated over a half dozen exits and more than$750 million in capital transactions including IPOs, secondary offerings, and debt instruments. -
Key opinion leader, Dr.
Giovanni Selvaggi , joinedCEL-SCI as a Clinical Advisor.Dr. Selvaggi , who has brought several oncology drugs to market, is a US-based drug developer, cancer researcher, and strategic advisor to big pharma and early-to-late-stage biotech companies. He joins several other top-tier physician consultants and head and neck cancer key opinion leaders who are advisors toCEL-SCI .
“We achieved significant and critical milestones in the third fiscal quarter including receiving the FDA’s green light on our confirmatory Registration Study of Multikine to address a vast unmet need in treating newly diagnosed head and neck cancer. Our data is so strong, and now further validated by the bias analysis, that the FDA has agreed to a 212-person study,” stated
Financial Results
Research and development expenses were
About
Multikine (Leukocyte Interleukin, Injection), a true first-line cancer therapy, has been dosed in over 740 patients and received Orphan Drug designation from the FDA for neoadjuvant therapy in patients with squamous cell carcinoma (cancer) of the head and neck. Multikine significantly extended life in its target patient population demonstrating a 73% survival rate with Multikine vs. only 45% without at 5 years after treatment. Based on this very strong data, the FDA agreed to CEL-SCI’s target patient selection criteria and gave the go-ahead to conduct a small, focused, confirmatory Registration Study which will enroll 212 patients.
The Company has operations in
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. When used in this press release, the words "intends," "believes," "anticipated," "plans" and "expects," and similar expressions, are intended to identify forward-looking statements. Such statements are subject to risks and uncertainties that could cause actual results to differ materially from those projected. Factors that could cause or contribute to such differences include an inability to duplicate the clinical results demonstrated in clinical studies, timely development of any potential products that can be shown to be safe and effective, receiving necessary regulatory approvals, difficulties in manufacturing any of the Company's potential products, inability to raise the necessary capital and the risk factors set forth from time to time in
* Multikine (Leukocyte Interleukin, Injection) is the trademark that
|
|
|||||||
|
CONDENSED STATEMENTS OF OPERATIONS |
|||||||
|
NINE MONTHS ENDED |
|||||||
|
(UNAUDITED) |
|||||||
|
|
|
|
|||||
|
2024 |
|
2023 |
|||||
|
Operating expenses: |
|
||||||
|
Research and development |
$ |
13,684,204 |
|
$ |
17,203,823 |
|
|
|
General and administrative |
|
6,547,866 |
|
|
6,804,729 |
|
|
|
Total operating expenses |
|
20,232,070 |
|
|
24,008,552 |
|
|
|
|
|
||||||
|
Operating loss |
|
(20,232,070 |
) |
|
(24,008,552 |
) |
|
|
Interest expense, net |
|
(571,102 |
) |
|
(493,522 |
) |
|
|
Other expense |
|
(7,801 |
) |
|
|
(61,525 |
) |
|
|
|
||||||
|
Net loss |
|
(20,810,973 |
) |
|
(24,563,599 |
) |
|
|
Modification of warrants |
|
(659,456 |
) |
|
(171,552 |
) |
|
|
|
|
||||||
|
Net loss available to common shareholders |
$ |
(21,470,429 |
) |
$ |
(24,735,151 |
) |
|
|
Net loss per common share – basic and diluted |
$ |
(0.42 |
) |
$ |
(0.57 |
) |
|
|
Weighted average common shares outstanding – basic and diluted |
|
51,479,454 |
|
|
43,761,395 |
|
|
|
|
|||||||
|
CONDENSED STATEMENTS OF OPERATIONS |
|||||||
|
THREE MONTHS ENDED |
|||||||
|
(UNAUDITED) |
|||||||
|
|
|
|
|||||
|
2024 |
|
2023 |
|||||
|
Operating expenses: |
|||||||
|
Research and development |
$ |
4,703,160 |
|
$ |
5,727,789 |
|
|
|
General and administrative |
|
1,967,075 |
|
|
2,453,968 |
|
|
|
Total operating expenses |
|
6,670,235 |
|
|
8,181,757 |
|
|
|
|
|
||||||
|
Operating loss |
|
(6,670,235 |
) |
|
(8,181,757 |
) |
|
|
Interest expense, net |
|
(190,705 |
) |
|
(181,670 |
) |
|
|
Other income (expense) |
|
4,382 |
|
|
|
(3,854 |
) |
|
|
|
||||||
|
Net loss |
|
(6,856,558 |
) |
|
(8,367,281 |
) |
|
|
Modification of warrants |
|
(659,456 |
) |
|
|
- |
|
|
|
|
||||||
|
Net loss available to common shareholders |
$ |
(7,516,014 |
) |
$ |
(8,367,281 |
) |
|
|
Net loss per common share – basic and diluted |
$ |
(0.14 |
) |
$ |
(0.19 |
) |
|
|
Weighted average common shares outstanding – basic and diluted |
|
53,994,384 |
|
|
44,254,363 |
|
|
View source version on businesswire.com: https://www.businesswire.com/news/home/20240815767286/en/
(703) 506-9460
Source:
