Johnson & Johnson Announces U.S. FDA Approval of TECVAYLI® plus DARZALEX FASPRO® for Relapsed/Refractory Multiple Myeloma, Offering a Potential New Standard of Care as Early as Second Line
Approval based on unprecedented Phase 3 data demonstrating statistically significant improvements in progression – free survival and overall survival versus standard of care regimens
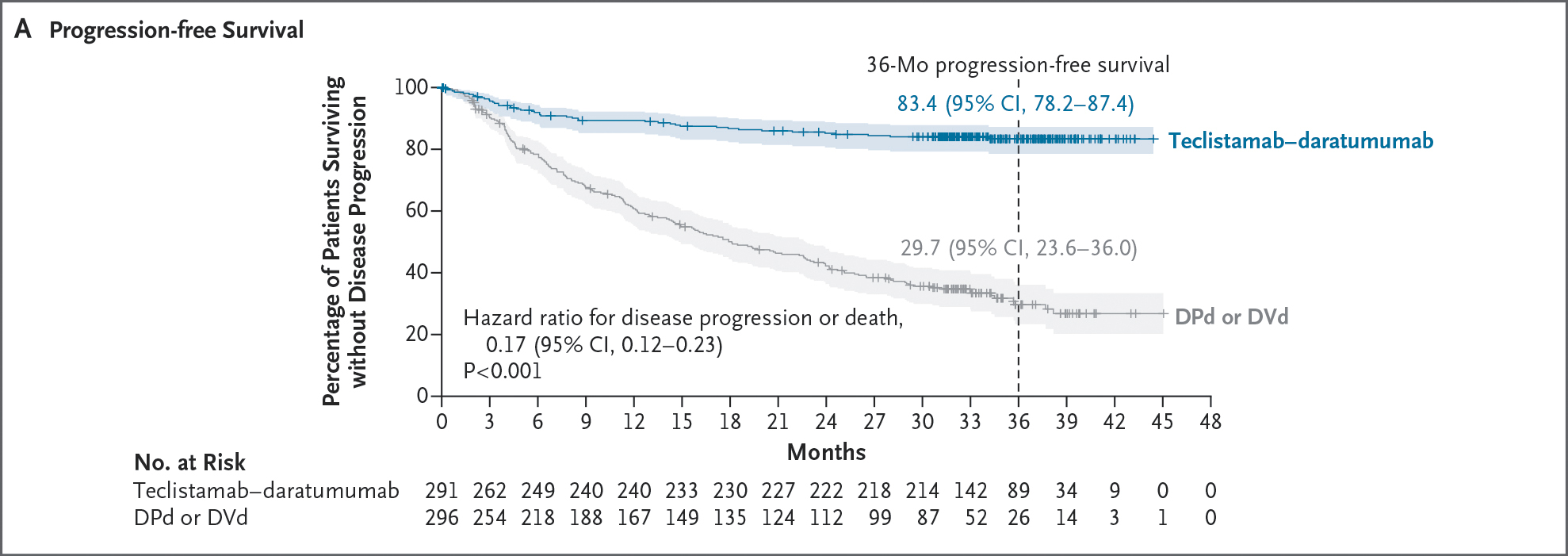
83.3% of patients were alive at three years, indicating durable clinical benefit
Multimedia assets for media are available here .
Perspectives on Expanding Multiple Myeloma Treatment Options
"This new treatment option can redefine how we approach RRMM treatment by giving healthcare providers a regimen with improvement in PFS and OS and a well-characterized safety profile," said Dr.
"There is a critical need to expand community-based treatment options for multiple myeloma patients, allowing them to receive care closer to home while respecting their individual treatment preferences," said
"As the leader in hematology, we have a proud history of transforming the treatment landscape for multiple myeloma. This approval represents another pivotal milestone in improving outcomes for patients living with this disease, with a unique regimen accessible to patients across all practice settings," said
Unprecedented MajesTEC-3 Study Results
The approval is based on data from the Phase 3 MajesTEC-3 study, an ongoing, Phase 3 randomized study evaluating the safety and efficacy of teclistamab plus daratumumab versus investigator's choice of daratumumab and dexamethasone with either pomalidomide or bortezomib in patients with RRMM who have received at least one prior line of therapy. TECVAYLI® in combination with DARZALEX
Significant improvements compared to SOC were observed across key secondary endpoints, including treatment response rates, minimal residual disease (MRD)-negativity, OS, and time to worsening of symptoms – revealing the impact of the regimen across varied patient measures.3 TECVAYLI® plus DARZALEX
In the MajesTEC-3 study, TECVAYLI® plus DARZALEX
The FDA proactively selected the teclistamab MajesTEC-3 supplemental Biologics License Application (sBLA) to participate in the Commissioner's National Priority Voucher (CNPV) Pilot Program as it aligns with the program's priority to deliver more innovative therapies for American people. The FDA also granted the application Breakthrough Therapy Designation and Real-Time Oncology Review.
Access to TECVAYLI® plus DARZ
ALEX
About MajesTEC-3
MajesTEC-3 is an ongoing, Phase 3 randomized study evaluating the safety and efficacy of teclistamab plus daratumumab versus investigator's choice of daratumumab and dexamethasone with either pomalidomide or bortezomib in patients with relapsed/refractory multiple myeloma who have received at least one prior line of therapy. The primary endpoint is PFS and secondary endpoints include complete response or better, overall response rate, minimal residual disease negativity, overall survival, time to worsening of symptoms (MySIm-Q), and safety. The MajesTEC-3 study is a part of the MajesTEC clinical program, which includes exploring the potential of TECVAYLI® as a combination regimen.4 It is the first randomized Phase 3 trial using a bispecific antibody in relapsed/refractory multiple myeloma and confirmatory trial after the initial FDA approval.
About TECVAYLI®
TECVAYLI® (teclistamab-cqyv) is a first-in-class, bispecific T-cell engager antibody therapy that uses innovative science to activate the immune system by binding to the CD3 receptor expressed on the surface of T-cells and to the B-cell maturation antigen (BCMA) expressed on the surface of multiple myeloma cells and some healthy B-lineage cells. TECVAYLI® received accelerated approval from the
In
The
For more information, visit www.TECVAYLI.com.
About DARZALEX
DARZALEX
DARZALEX® (daratumumab) received
DARZALEX® is the first CD38-directed antibody approved to treat multiple myeloma.6 DARZALEX®-based regimens have been used in the treatment of more than 618,000 patients worldwide and more than 68,000 patients in the
In
For more information, visit www.DARZALEX.com.
About Multiple Myeloma
Multiple myeloma is an incurable blood cancer that affects a type of white blood cell called plasma cells, which are found in the bone marrow.8 In multiple myeloma, these plasma cells proliferate and spread rapidly and replace normal cells in the bone marrow with tumors.9 Multiple myeloma is the third most common blood cancer worldwide and remains an incurable disease.10 In 2024, it was estimated that more than 35,000 people will be diagnosed with multiple myeloma in the
TECVAYLI® INDICATIONS AND IMPORTANT SAFETY INFORMATION
INDICATIONS AND USAGE
TECVAYLI® (teclistamab-cqyv) is a bispecific B-cell maturation antigen (BCMA)-directed CD3 T-cell engager indicated for the treatment of adult patients with relapsed or refractory multiple myeloma:
- in combination with daratumumab and hyaluronidase-fihj in patients who have received at least one prior line of therapy, including a proteasome inhibitor and an immunomodulatory agent.
- as monotherapy, in patients who have received at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody.
IMPORTANT SAFETY INFORMATION
WARNING: CYTOKINE RELEASE SYNDROME and NEUROLOGIC TOXICITY including IMMUNE EFFECTOR CELL-ASSOCIATED NEUROTOXICITY SYNDROME
Cytokine release syndrome (CRS), including life-threatening or fatal reactions, can occur in patients receiving TECVAYLI. Initiate treatment with TECVAYLI step-up dosing schedule to reduce risk of CRS. Withhold TECVAYLI until CRS resolves or permanently discontinue based on severity.
Neurologic toxicity, including Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) and serious, life-threatening, or fatal reactions, can occur in patients receiving TECVAYLI. Monitor patients for signs or symptoms of neurologic toxicity, including ICANS, during treatment. Withhold TECVAYLI until neurologic toxicity resolves or permanently discontinue based on severity.
TECVAYLI is available only through a restricted program called the TECVAYLI and TALVEY® Risk Evaluation and Mitigation Strategy (REMS).
WARNINGS AND PRECAUTIONS
Cytokine Release Syndrome - TECVAYLI can cause cytokine release syndrome (CRS), including life-threatening or fatal reactions.
In the clinical trials (monotherapy and combination therapy; N=448), CRS occurred in 64% of patients who received TECVAYLI at the recommended dose, with Grade 1 CRS occurring in 46% of patients, Grade 2 in 18%, and Grade 3 in 0.2%. Recurrent CRS occurred in 27% of patients. Most patients experienced CRS during the initial step-up dosing schedule (step-up dose 1 [37%], step-up dose 2 [32%], or the initial treatment dose [20%]). CRS first occurred following subsequent doses of TECVAYLI in 2.5% of patients. The median time to onset of CRS was 2 (range: 1 to 9) days after the most recent dose and the median duration of CRS was 2 (range: 1 to 22) days.
Clinical signs and symptoms of CRS included, but were not limited to, fever, hypoxia, chills, hypotension, sinus tachycardia, headache, and elevated liver enzymes (aspartate aminotransferase and alanine aminotransferase elevation).
Initiate therapy according to TECVAYLI step-up dosing schedule to reduce risk of CRS. Administer pretreatment medications to reduce risk of CRS and monitor patients following administration of TECVAYLI accordingly.
At the first sign of CRS, immediately evaluate patient for hospitalization. Administer supportive care based on severity and consider further management per current practice guidelines. Withhold until CRS resolves or permanently discontinue TECVAYLI based on severity.
TECVAYLI is available only through a restricted program under a REMS.
Neurologic Toxicity including Immune Effector Cell-Associated Neurotoxicity Syndrome - TECVAYLI can cause serious, life-threatening, or fatal neurologic toxicity, including immune effector cell-associated neurotoxicity syndrome (ICANS).
In the clinical trials (monotherapy and combination therapy trials; N=448), neurologic toxicity occurred in 60% of patients who received TECVAYLI at the recommended dosage, with Grade 3 or 4 neurologic toxicity in 6%. Neurologic toxicities reported in ≥5% of patients included headache (27%), sensory neuropathy (16%), motor dysfunction (15%), insomnia (12%), encephalopathy (11%), and dizziness (8%). Fatal neurologic toxicity occurred in 0.4% of patients, including Guillain-Barré syndrome and status epilepticus (one patient each).
In MajesTEC-1, ICANS was reported in 6% of patients who received TECVAYLI as monotherapy at the recommended dosage. Recurrent ICANS occurred in 1.8% of patients. Most patients experienced ICANS following step-up dose 1 (1.2%), step-up dose 2 (0.6%), or the initial treatment dose (1.8%). Less than 3% of patients developed first occurrence of ICANS following subsequent TECVAYLI doses. The median time to onset of ICANS was 4 (range: 2 to 8) days after the most recent dose with a median duration of 3 (range: 1 to 20) days. The most frequent clinical manifestations of ICANS reported were confusional state and dysgraphia.
In MajesTEC-3, ICANS was reported in 1.1% of patients who received the recommended TECVAYLI dosage in combination with daratumumab and hyaluronidase-fihj, including Grade 4 ICANS in 1 patient. All events of ICANS occurred during the step-up dosing schedule. The median time to onset of ICANS was 2 (range: 1 to 3) days after the most recent dose and the median duration of ICANS was 2 (range: 1 to 2) days. The clinical manifestations of ICANS reported were amnesia, encephalopathy and delirium.
The onset of ICANS can be concurrent with CRS, following resolution of CRS, or in the absence of CRS.
Monitor patients for signs and symptoms of neurologic toxicity, including ICANS during TECVAYLI treatment. At the first sign of neurologic toxicity, including ICANS, immediately evaluate patient and provide supportive therapy based on severity. Withhold until neurologic toxicity resolves or permanently discontinue TECVAYLI based on severity per recommendations and consider further management per current practice guidelines.
Due to the potential for neurologic toxicity, patients receiving TECVAYLI are at risk of depressed level of consciousness. Advise patients to refrain from driving or operating heavy or potentially dangerous machinery during and for 48 hours after completion of TECVAYLI step-up dosing schedule and in the event of new onset of any neurologic toxicity symptoms until neurologic toxicity resolves.
TECVAYLI is available only through a restricted program under a REMS.
TECVAYLI and TALVEY REMS - TECVAYLI is available only through a restricted program under a REMS called the TECVAYLI and TALVEY REMS because of the risks of CRS and neurologic toxicity, including ICANS.
Hepatotoxicity - TECVAYLI can cause hepatotoxicity, including fatalities. There was one fatal case of hepatic failure in MajesTEC-1. In patients who received TECVAYLI at the recommended dose in the clinical trials (monotherapy and combination therapy trials; N=448) elevated aspartate aminotransferase (AST) occurred in 47% of patients, with Grade 3 or 4 elevations in 2.9%. Elevated alanine aminotransferase (ALT) occurred in 48% of patients, with Grade 3 or 4 elevations in 3.8%. Elevated total bilirubin occurred in 10% of patients with Grade 3 or 4 elevations in 0.7%. Liver enzyme elevation can occur with or without concurrent CRS.
Monitor liver enzymes and bilirubin at baseline and during treatment as clinically indicated. Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity.
Infections - TECVAYLI can cause severe, life-threatening, or fatal infections.
In MajesTEC-1 (N=165), in patients who received the recommended TECVAYLI dosage, serious infections, including opportunistic infections, occurred in 30% of patients, Grade 3 or 4 infections in 35% of patients, and fatal infections in 4.2% of patients.
In MajesTEC-3 (N=283), in patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj at the recommended dosage, serious infections, including opportunistic infections, occurred in 54% of patients, Grade 3 or Grade 4 infections in 54% of patients, and fatal infections in 4.6% of patients.
Monitor patients for signs and symptoms of infection prior to and during treatment with TECVAYLI and treat appropriately. Administer prophylactic antimicrobials according to current practice guidelines.
Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity.
Monitor immunoglobulin levels prior to and during treatment with TECVAYLI and administer subcutaneous or intravenous immunoglobulin (IVIG) to maintain the serum levels >400 mg/dL.
Neutropenia - TECVAYLI can cause neutropenia and febrile neutropenia. In patients who received TECVAYLI at the recommended dose in the clinical trials (monotherapy and combination therapy trials; N=448), decreased neutrophils occurred in 88% of patients, with Grade 3 or 4 decreased neutrophils in 70%. Febrile neutropenia occurred in 6% of patients.
Monitor complete blood cell counts at baseline and periodically during treatment and provide supportive care per local institutional guidelines.
Monitor patients with neutropenia for signs of infection.
Withhold TECVAYLI based on severity.
Hypersensitivity and Other Administration Reactions - TECVAYLI can cause both systemic administration-related and local injection-site reactions.
Systemic Reactions - In patients who received the recommended TECVAYLI dosage in the clinical trials (monotherapy and combination therapy trials; N=448), 2.5% of patients experienced systemic-administration reactions, which included recurrent pyrexia and rash.
Local Reactions - In patients who received TECVAYLI at the recommended dosage in the clinical trials (monotherapy and combination therapy trials; N=448), injection-site reactions occurred in 37% of patients, with Grade 1 injection-site reactions in 29% and Grade 2 in 9%.
Withhold TECVAYLI or consider permanent discontinuation of TECVAYLI based on severity.
Embryo-Fetal Toxicity - Based on its mechanism of action, TECVAYLI may cause fetal harm when administered to a pregnant patient. Advise pregnant patients of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with TECVAYLI and for 5 months after the last dose.
ADVERSE REACTIONS
The most common adverse reactions (≥20%) in patients who received TECVAYLI monotherapy were pyrexia, CRS, musculoskeletal pain, injection site reaction, fatigue, upper respiratory tract infection, nausea, headache, pneumonia, and diarrhea. The most common adverse reactions (≥20%) in patients who received TECVAYLI in combination with daratumumab and hyaluronidase-fihj were hypogammaglobulinemia, upper respiratory tract infection, CRS, cough, diarrhea, musculoskeletal pain, COVID-19, pneumonia, injection site reaction, fatigue, pyrexia, headache, nausea, gastroenteritis, and weight decreased.
The most common Grade 3 to 4 laboratory abnormalities (≥20%) with TECVAYLI (as monotherapy or in combination with daratumumab and hyaluronidase-fihj) were decreased lymphocytes, decreased neutrophils, decreased white blood cells, decreased hemoglobin, and decreased platelets.
Please read full Prescribing Information , including Boxed WARNING, for TECVAYLI.
cp-322928v6
DARZALEX
INDICATIONS
DARZALEX
- In combination with bortezomib, lenalidomide, and dexamethasone for induction and consolidation in newly diagnosed patients who are eligible for autologous stem cell transplant
- In combination with bortezomib, lenalidomide, and dexamethasone in newly diagnosed patients who are ineligible for autologous stem cell transplant
- In combination with bortezomib, melphalan, and prednisone in newly diagnosed patients who are ineligible for autologous stem cell transplant
- In combination with lenalidomide and dexamethasone in newly diagnosed patients who are ineligible for autologous stem cell transplant and in patients with relapsed or refractory multiple myeloma who have received at least one prior therapy
- In combination with bortezomib, thalidomide, and dexamethasone in newly diagnosed patients who are eligible for autologous stem cell transplant
- In combination with pomalidomide and dexamethasone in patients who have received at least one prior line of therapy including lenalidomide and a proteasome inhibitor (PI)
- In combination with carfilzomib and dexamethasone in patients with relapsed or refractory multiple myeloma who have received one to three prior lines of therapy
- In combination with bortezomib and dexamethasone in patients who have received at least one prior therapy
- As monotherapy in patients who have received at least three prior lines of therapy including a PI and an immunomodulatory agent or who are double refractory to a PI and an immunomodulatory agent
DARZALEX
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
DARZALEX
WARNINGS AND PRECAUTIONS
Hypersensitivity and Other Administration Reactions
Both systemic administration-related reactions, including severe or life-threatening reactions, and local injection-site reactions can occur with DARZALEX
Systemic Reactions
In a pooled safety population of 1446 patients with multiple myeloma (N=1235) or light chain (AL) amyloidosis (N=193) who received DARZALEX
In all patients (N=1639), systemic administration-related reactions occurred in 7% of patients with the first injection, 0.5% with the second injection, and cumulatively 1% with subsequent injections. The median time to onset was 3.2 hours (range: 4 minutes to 3.5 days). Of the 283 systemic administration-related reactions that occurred in 135 patients, 240 (85%) occurred on the day of DARZALEX
Severe reactions included hypoxia, dyspnea, hypertension, tachycardia, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Other signs and symptoms of systemic administration-related reactions may include respiratory symptoms, such as bronchospasm, nasal congestion, cough, throat irritation, allergic rhinitis, and wheezing, as well as anaphylactic reaction, pyrexia, chest pain, pruritus, chills, vomiting, nausea, hypotension, and blurred vision.
Pre-medicate patients with histamine-1 receptor antagonist, acetaminophen, and corticosteroids. Monitor patients for systemic administration-related reactions, especially following the first and second injections. For anaphylactic reaction or life-threatening (Grade 4) administration-related reactions, immediately and permanently discontinue DARZALEX
Ocular adverse reactions, including acute myopia and narrowing of the anterior chamber angle due to ciliochoroidal effusions with potential for increased intraocular pressure or glaucoma, have occurred with daratumumab-containing products. If ocular symptoms occur, interrupt DARZALEX
Local Reactions
In this pooled safety population of 1446 patients with multiple myeloma (N=1253) or light chain amyloidosis (N=193), injection-site reactions occurred in 8% of patients, including Grade 2 reactions in 1.1%. The most frequent (>1%) injection-site reactions were injection site erythema and injection site rash. In patients with high-risk smoldering multiple myeloma (N=193), injection-site reactions occurred in 28% of patients, including Grade 2 reactions in 3%. These local reactions occurred a median of 6 minutes (range: 0 minutes to 6.5 days) after starting administration of DARZALEX
Infections
DARZALEX
Monitor patients for signs and symptoms of infection prior to and during treatment with DARZALEX
Neutropenia
Daratumumab may increase neutropenia induced by background therapy. Monitor complete blood cell counts periodically during treatment according to manufacturer's prescribing information for background therapies. Monitor patients with neutropenia for signs of infection. Consider withholding DARZALEX
Thrombocytopenia
Daratumumab may increase thrombocytopenia induced by background therapy. Monitor complete blood cell counts periodically during treatment according to manufacturer's prescribing information for background therapies. Consider withholding DARZALEX
Embryo-Fetal Toxicity
Based on the mechanism of action, DARZALEX
The combination of DARZALEX
Interference With Serological Testing
Daratumumab binds to CD38 on red blood cells (RBCs) and results in a positive indirect antiglobulin test (indirect Coombs test). Daratumumab-mediated positive indirect antiglobulin test may persist for up to 6 months after the last daratumumab administration. Daratumumab bound to RBCs masks detection of antibodies to minor antigens in the patient's serum. The determination of a patient's ABO and Rh blood type are not impacted.
Notify blood transfusion centers of this interference with serological testing and inform blood banks that a patient has received DARZALEX
Interference With Determination of Complete Response
Daratumumab is a human immunoglobulin G (IgG) kappa monoclonal antibody that can be detected on both the serum protein electrophoresis (SPE) and immunofixation (IFE) assays used for the clinical monitoring of endogenous M-protein. This interference can impact the determination of complete response and of disease progression in some DARZALEX
ADVERSE REACTIONS
In multiple myeloma, the most common adverse reaction (≥20%) with DARZALEX
The most common adverse reactions (≥20%) in patients with high-risk smoldering multiple myeloma who received DARZALEX
The most common hematology laboratory abnormalities (≥40%) with DARZALEX
Please
click here
to read full Prescribing Information for DARZALEX
DARZALEX® INDICATIONS AND IMPORTANT SAFETY INFORMATION
INDICATIONS
DARZALEX® (daratumumab) is indicated for the treatment of adult patients with multiple myeloma:
- In combination with bortezomib, melphalan, and prednisone in newly diagnosed patients who are ineligible for autologous stem cell transplant
- In combination with lenalidomide and dexamethasone in newly diagnosed patients who are ineligible for autologous stem cell transplant and in patients with relapsed or refractory multiple myeloma who have received at least one prior therapy
- In combination with bortezomib, thalidomide, and dexamethasone in newly diagnosed patients who are eligible for autologous stem cell transplant
- In combination with pomalidomide and dexamethasone in patients who have received at least one prior line of therapy including lenalidomide and a proteasome inhibitor
- In combination with carfilzomib and dexamethasone in patients with relapsed or refractory multiple myeloma who have received one to three prior lines of therapy
- In combination with bortezomib and dexamethasone in patients who have received at least one prior therapy
- As monotherapy in patients who have received at least three prior lines of therapy including a proteasome inhibitor (PI) and an immunomodulatory agent or who are double-refractory to a PI and an immunomodulatory agent
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
DARZALEX® is contraindicated in patients with a history of severe hypersensitivity (eg, anaphylactic reactions) to daratumumab or any of the components of the formulation.
WARNINGS AND PRECAUTIONS
Infusion-Related Reactions
DARZALEX® can cause severe and/or serious infusion-related reactions including anaphylactic reactions. These reactions can be life threatening, and fatal outcomes have been reported. In clinical trials (monotherapy and combination: N=2066), infusion-related reactions occurred in 37% of patients with the Week 1 (16 mg/kg) infusion, 2% with the Week 2 infusion, and cumulatively 6% with subsequent infusions. Less than 1% of patients had a Grade 3/4 infusion-related reaction at Week 2 or subsequent infusions. The median time to onset was 1.5 hours (range: 0 to 73 hours). Nearly all reactions occurred during infusion or within 4 hours of completing DARZALEX®. Severe reactions have occurred, including bronchospasm, hypoxia, dyspnea, hypertension, tachycardia, headache, laryngeal edema, pulmonary edema, and ocular adverse reactions, including choroidal effusion, acute myopia, and acute angle closure glaucoma. Signs and symptoms may include respiratory symptoms, such as nasal congestion, cough, throat irritation, as well as chills, vomiting, and nausea. Less common signs and symptoms were wheezing, allergic rhinitis, pyrexia, chest discomfort, pruritus, hypotension and blurred vision.
When DARZALEX® dosing was interrupted in the setting of ASCT (CASSIOPEIA) for a median of 3.75 months (range: 2.4 to 6.9 months), upon re-initiation of DARZALEX®, the incidence of infusion-related reactions was 11% for the first infusion following ASCT. Infusion-related reactions occurring at re-initiation of DARZALEX® following ASCT were consistent in terms of symptoms and severity (Grade 3 or 4: <1%) with those reported in previous studies at Week 2 or subsequent infusions. In EQUULEUS, patients receiving combination treatment (n=97) were administered the first 16 mg/kg dose at Week 1 split over two days, ie, 8 mg/kg on Day 1 and Day 2, respectively. The incidence of any grade infusion-related reactions was 42%, with 36% of patients experiencing infusion-related reactions on Day 1 of Week 1, 4% on Day 2 of Week 1, and 8% with subsequent infusions.
Pre-medicate patients with antihistamines, antipyretics, and corticosteroids. Frequently monitor patients during the entire infusion. Interrupt DARZALEX® infusion for reactions of any severity and institute medical management as needed. Permanently discontinue DARZALEX® therapy if an anaphylactic reaction or life-threatening (Grade 4) reaction occurs and institute appropriate emergency care. For patients with Grade 1, 2, or 3 reactions, reduce the infusion rate when re-starting the infusion.
To reduce the risk of delayed infusion-related reactions, administer oral corticosteroids to all patients following DARZALEX® infusions. Patients with a history of chronic obstructive pulmonary disease may require additional post-infusion medications to manage respiratory complications. Consider prescribing short- and long-acting bronchodilators and inhaled corticosteroids for patients with chronic obstructive pulmonary disease.
Ocular adverse reactions, including acute myopia and narrowing of the anterior chamber angle due to ciliochoroidal effusions with potential for increased intraocular pressure or glaucoma, have occurred with DARZALEX® infusion. If ocular symptoms occur, interrupt DARZALEX® infusion and seek immediate ophthalmologic evaluation prior to restarting DARZALEX®.
Interference With Serological Testing
Daratumumab binds to CD38 on red blood cells (RBCs) and results in a positive indirect antiglobulin test (indirect Coombs test). Daratumumab-mediated positive indirect antiglobulin test may persist for up to 6 months after the last daratumumab infusion. Daratumumab bound to RBCs masks detection of antibodies to minor antigens in the patient's serum. The determination of a patient's ABO and Rh blood type is not impacted. Notify blood transfusion centers of this interference with serological testing and inform blood banks that a patient has received DARZALEX®. Type and screen patients prior to starting DARZALEX®.
Neutropenia and Thrombocytopenia
DARZALEX® may increase neutropenia and thrombocytopenia induced by background therapy. Monitor complete blood cell counts periodically during treatment according to manufacturer's prescribing information for background therapies. Monitor patients with neutropenia for signs of infection. Consider withholding DARZALEX® until recovery of neutrophils or for recovery of platelets.
Interference With Determination of Complete Response
Daratumumab is a human immunoglobulin G (IgG) kappa monoclonal antibody that can be detected on both the serum protein electrophoresis (SPE) and immunofixation (IFE) assays used for the clinical monitoring of endogenous M-protein. This interference can impact the determination of complete response and of disease progression in some patients with IgG kappa myeloma protein.
Embryo-Fetal Toxicity
Based on the mechanism of action, DARZALEX® can cause fetal harm when administered to a pregnant woman. DARZALEX® may cause depletion of fetal immune cells and decreased bone density. Advise pregnant women of the potential risk to a fetus. Advise females with reproductive potential to use effective contraception during treatment with DARZALEX® and for 3 months after the last dose.
The combination of DARZALEX® with lenalidomide, pomalidomide, or thalidomide is contraindicated in pregnant women because lenalidomide, pomalidomide, and thalidomide may cause birth defects and death of the unborn child. Refer to the lenalidomide, pomalidomide, or thalidomide prescribing information on use during pregnancy.
ADVERSE REACTIONS
The most frequently reported adverse reactions (incidence ≥20%) were upper respiratory infection, neutropenia, infusion-related reactions, thrombocytopenia, diarrhea, constipation, anemia, peripheral sensory neuropathy, fatigue, peripheral edema, nausea, cough, pyrexia, dyspnea, and asthenia. The most common hematologic laboratory abnormalities (≥40%) with DARZALEX® are neutropenia, lymphopenia, thrombocytopenia, leukopenia, and anemia.
Please click here to see the full Prescribing Information.
About Johnson & Johnson
At Johnson & Johnson, we believe health is everything. Our strength in healthcare innovation empowers us to build a world where complex diseases are prevented, treated, and cured, where treatments are smarter and less invasive, and solutions are personal. Through our expertise in Innovative Medicine and MedTech, we are uniquely positioned to innovate across the full spectrum of healthcare solutions today to deliver the breakthroughs of tomorrow, and profoundly impact health for humanity.
Learn more at https://www.jnj.com/ or at www.innovativemedicine.jnj.com.
Cautions Concerning Forward-Looking Statements
This press release contains "forward-looking statements" as defined in the Private Securities Litigation Reform Act of 1995 regarding product development and the potential benefits and treatment impact of TECVAYLI® (teclistamab-cqyv) and DARZALEX FASPRO® (daratumumab and hyaluronidase-fihj). The reader is cautioned not to rely on these forward-looking statements. These statements are based on current expectations of future events. If underlying assumptions prove inaccurate or known or unknown risks or uncertainties materialize, actual results could vary materially from the expectations and projections of
FOOTNOTES
*
Dr.
**
†
The patient support and resources provided by J&J withMe are not intended to give medical advice, replace a treatment plan from the patient's healthcare provider, offer services that would normally be performed by the provider's office, or serve as a reason to prescribe a
References
1 TECVAYLI®
2 Kumar, S. K., et al. (2021). Prognostic factors for early (<2 years) and late (>2 years) relapse after initial therapy in multiple myeloma. Blood, 138(Supplement 1), 3761. https://doi.org/10.1182/blood-2021-151249
3
4 MajesTEC-3, NCT05083169. A Phase 3 Randomized Study Comparing Teclistamab + Subcutaneous Daratumumab (Tec-Dara) Versus Daratumumab SC + Pomalidomide + Dexamethasone (DPd) or Daratumumab SC + Bortezomib + Dexamethasone (DVd). https://clinicaltrials.gov/study/NCT05083169. Accessed
5
6 DARZALEX
7 DARZALEX®
8 Rajkumar SV. Multiple Myeloma: 2020 Update on Diagnosis, Risk-Stratification and Management. Am J Hematol. 2020;95(5):548-567. http://www.ncbi.nlm.nih.gov/pubmed/32212178.
9
10
11
12 SEER Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance Research Program,
13
14

https://clinicaltrials.gov/study/NCT05083169
SOURCE
